Projects
Transparent ECoG
2018
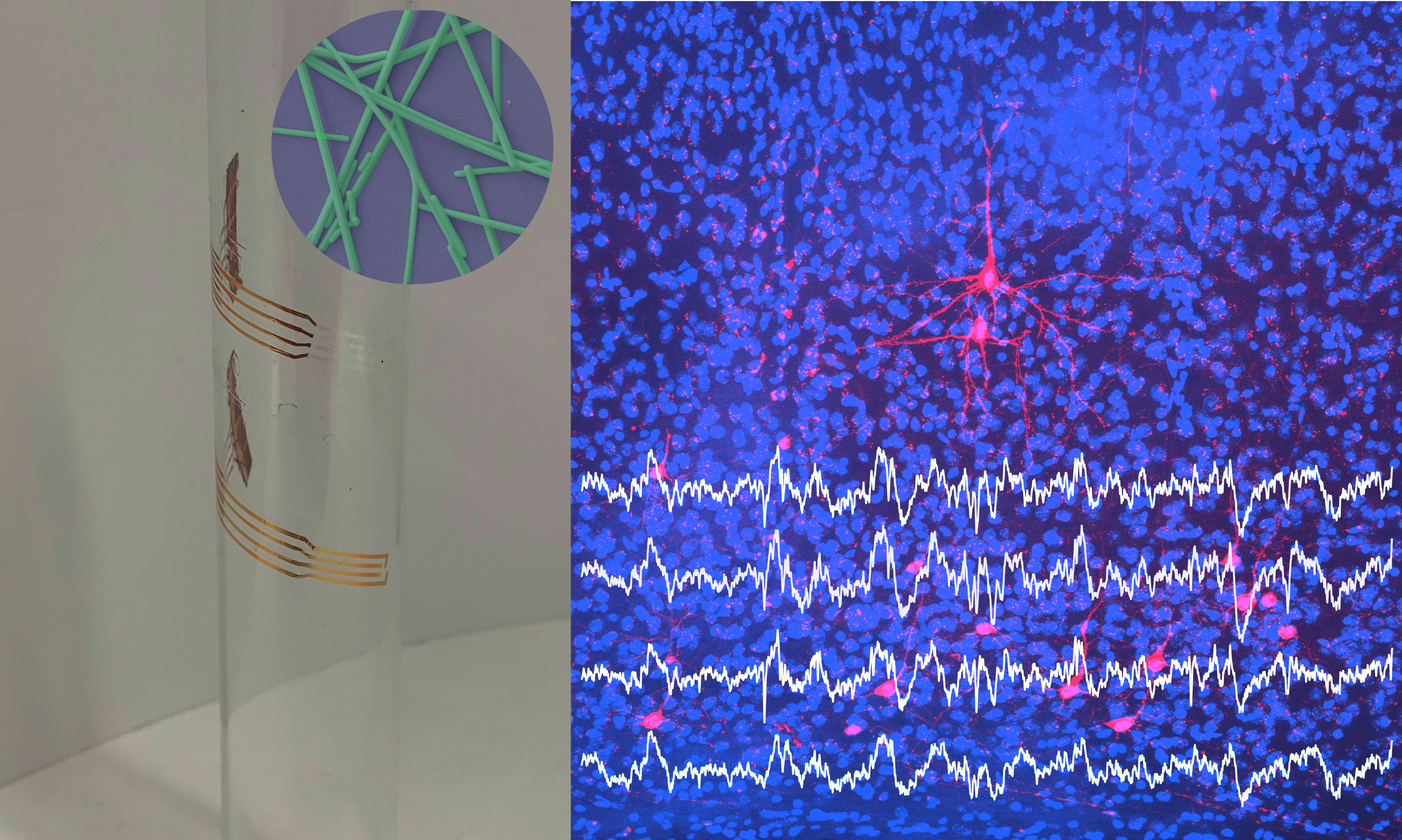
In 2018 I have started to explore, as PosDoctoral Research Scientist in Sainsbury Welcome Centre and CENIMAT/I3N, new transparent materials for extracellular electrodes. The goal of this project is to fabricate large-scale, high-density transparent and flexible electrode arrays, that combine the superior temporal resolution of extracellular electrophysiology with the spatial resolution offered by optical imaging. This project will explore the electrical, optical, and mechanical properties of emerging transparent conductive materials for developing high-performance and low-cost electrocorticography (ECoG) electrode arrays. The validation of new transparent ECoG will be performed in vivo by combining extracellular recordings with functional calcium imaging. Currently, I’m co-supervising with Hugo Marques from Champalimaud Neuroscience Program, a master student working on ‘Transparent and flexible ECoG electrode arrays of metallic nanostructures for neural recordings’. I’m also involved in teaching undergraduate students about the fabrication and characterization of transparent and conductive films at CENIMAT/I3N.
Ultra-thin and conformable Parylene-based circuitry
2018

Parylene membranes (1 to 5 µm thick) are biocompatible, transparent and compatible with standard microfabrication processes (e.g., photolitography). At CENIMAT/I3N, in collaboration with Professor Joana Vaz Pinto, we were able to fabricate on ultra-thin Parylene membranes- TFTs, MIM, Resistors, Capacitors, temperature and UV sensors, as well as RFID antennas. We co-supervise several master students working on this subject - (1)‘Development of temperature sensor arrays in ultrathin membranes of Parylene’, (2)‘Development of of Temperature sensors in Parylene membranes’, (3)‘Integration of IC temperature sensors in conformable membranes of Parylene’, (4)‘Fabrication of RFID antennas in Parylene membranes for wearables’, and (5)‘Fabrication of UV sensors in Parylene membranes for wearables.’
Microfluidics
2019
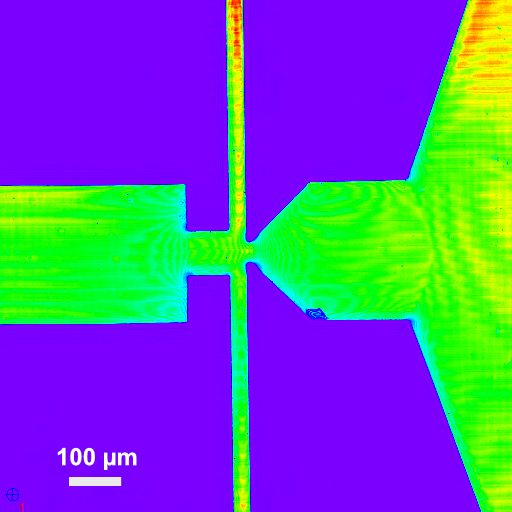
In 2019, I’ve started to work in this project as Posdoctoral Research Associate at CENIMAT/I3N. The project involves fabricating a microfluidic device for the rapid detection of cancer from circulating free DNA in blood and urine. For effectively addressing the challenges present in this project we must combine different materials and microfabrication technologies, such as direct laser writing (DLW). This novel technology enables a fast fabrication of precise and complex microstructures, which are much more difficult and time demanding to fabricate with standard patterning technologies, like photolithography. Recently, I won a Yerun Research Mobility Award to visit Vellekoop Group at Bermen University to explore and increase my expertise on this technology. Currently, I’m supervising a master student working on the ‘Development of a Microfluidic Device for DNA amplification using Digital PCR.’ Moreover, I’m teaching undergraduate students about colorimetric DNA detection using microfluidics at CENIMAT/I3N.
Interfacing Materials and Neuroscience
2012

This was my PhD project. Extracellular recording remains the only technique capable of measuring the activity of many neurons simultaneously with a sub-millisecond precision, in multiple brain areas, including deep structures. Nevertheless, many questions about the nature of the detected signal and the limitations/capabilities of this technique remain unanswered.
“Can we improve data quality by electrodes design choice (e.g., impedance and size)? How will these be reflected in the subsequent sorting analysis? What arrangement of electrodes is optimal for isolating individual neurons from background noise? How can we validate probes with different electrode configurations and different sorting algorithms? How can we achieve the full potential of large-scale, high-density extracellular recordings?”
For understanding and improving neural probes, we provided new methods to quantify their performance, as well as new validation datasets. All data was made freely available online. These datasets have been used by several groups, contributing to the advancement of knowledge on spike sorting algorithms. Moreover, extracellular neural probes have made a big step towards the large-scale, high-density extracellular recordings. In collaboration with the European Project NeuroSeeker, we validated a CMOS-based probe with 1344 electrodes allowing the simultaneous recording of more than 1000 neurons spanning different brain regions. In collaboration with Gonçalo Lopes and George Dimitriadis, we presented new methods for visualizing and analysing big neural data. My PhD work has also contributed to attract national funds for the project ‘Interfacing Technology with the Brain: Novel materials for implantable neural devices’ (BIAL Foundation grant). Additionally, outreach activities were realized where we shared the most recent technological advances using CMOS-based probes (‘Orchestrating the brain’ at Sainsbury Welcome Centre). During my PhD I was also involved in teaching and mentoring undergrads and master students, who have collaborated on my work.
Ph Sensor
2014
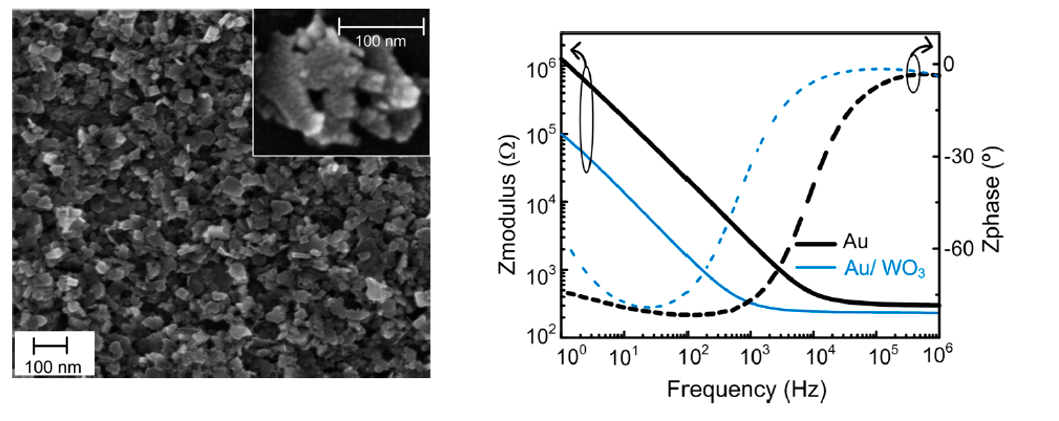
During my PhD, I explored the electrodeposition thecnique at Cenimat/I3N, showing that was possible to reliable coat metal electrodes with different materials (e.g. WO3 and PEDOT-PSS). In collaboration with Lídia Santos, we fabricated WO3 nanoparticle-based conformable pH sensors. These sensors shown a sensitivity of −56.7 ± 1.3 mV/pH, in a wide pH range of 9 to 5. A good balance between sensitivity (theoretical maximum sensitivity is −59 mV/pH), production costs, and simplicity of the sensors was achieved. Besides, this metal oxide-based pH sensor, where pH sensing is achieved through a potentiometric method, I’ve also explored capacitive field-effect pH sensors based on an electrolyte–insulator–semiconductor (EIS) structure. At CENIMAT/I3N, I taught undergrads students about field effect based sensors - ion sensitive field-effect transistors (ISFET) and EIS capacitors.
Biobatteries
2010
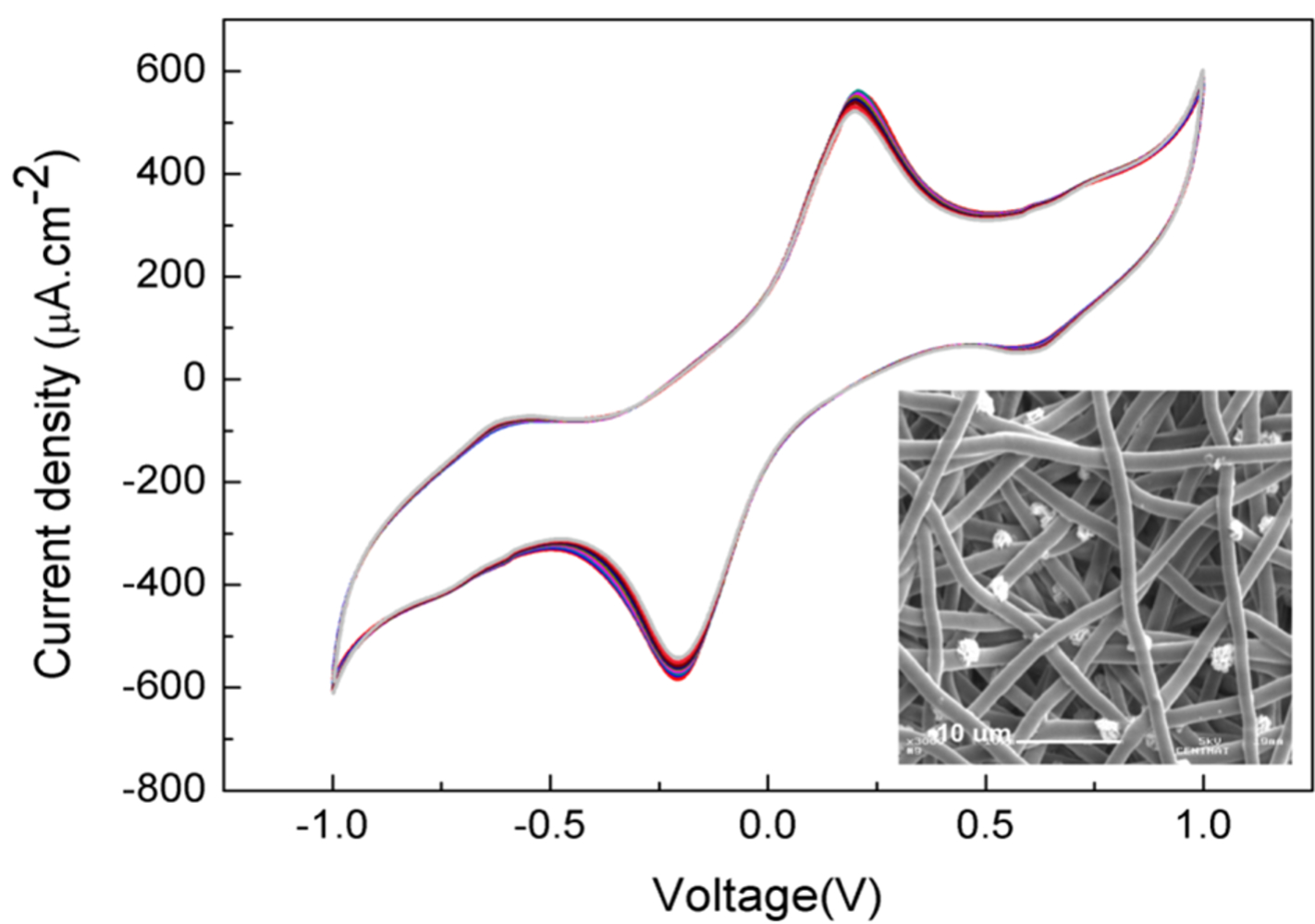
This was my MSc project. A liquid-activated power source based on a flexible and thin electrospun polycaprolactone (PCL) membrane is designed for harvesting energy when in contact with biofluids, targeting healthcare energy powering applications. The PCL membrane separates two electrodes: an oxidizing electrode (anode) and a reducing electrode (cathode). Moreover, the PCL membrane contributes to the efficiency of the charge generation processes that occur at the electrodes and the charge transport across the electrolyte. The presence of biofluids leads to a stable and reversible electrochemical behavior (redox reactions at terminal C-OH groups of the microfibers at ± 0.18 V) that is highly dependent on the surface area and the interconnected pore structure of the PCL electrospun membrane. This project was awarded by the European Materials Society.
